Metabolic Blindness: How "Healthy" Eating Triggers Blood Glucose Spikes and Accelerates Aging
Seemingly healthy foods trigger acute blood glucose spikes in non-diabetic individuals. Learn how chronic glucose variability impairs brain function, accelerates aging, and what you can do about it.
The Evidence
Seemingly healthy foods—whole-grain oatmeal, whole-wheat bread, fresh fruit juice—trigger acute blood glucose spikes (40-60 mg/dL within 15-30 minutes) in non-diabetic individuals. Chronic glucose variability impairs brain glucose uptake despite elevated blood glucose (the "starving brain" paradox), accelerates skin aging through Advanced Glycation End-products (AGEs), and increases cardiovascular mortality risk by 3-5% per mmol/L increase in postprandial glucose. Continuous Glucose Monitoring (CGM) enables personalized metabolic optimization and can reduce glucose variability by 35-40% within 4 weeks.
Introduction: The Health Paradox
The modern health-conscious consumer faces a paradox. You follow conventional nutritional guidance: whole grains instead of refined carbohydrates, fresh fruit instead of candy, whole-wheat bread instead of white bread. Yet despite these choices, you experience persistent afternoon fatigue, difficulty concentrating, and unexplained weight gain.
This article examines the scientific evidence behind this paradox. We synthesize findings from 10 peer-reviewed studies across endocrinology, neurology, and gerontology to explain why conventional dietary recommendations may inadvertently trigger metabolic dysfunction in otherwise healthy individuals.
Part 1: Understanding Postprandial Glucose Dynamics
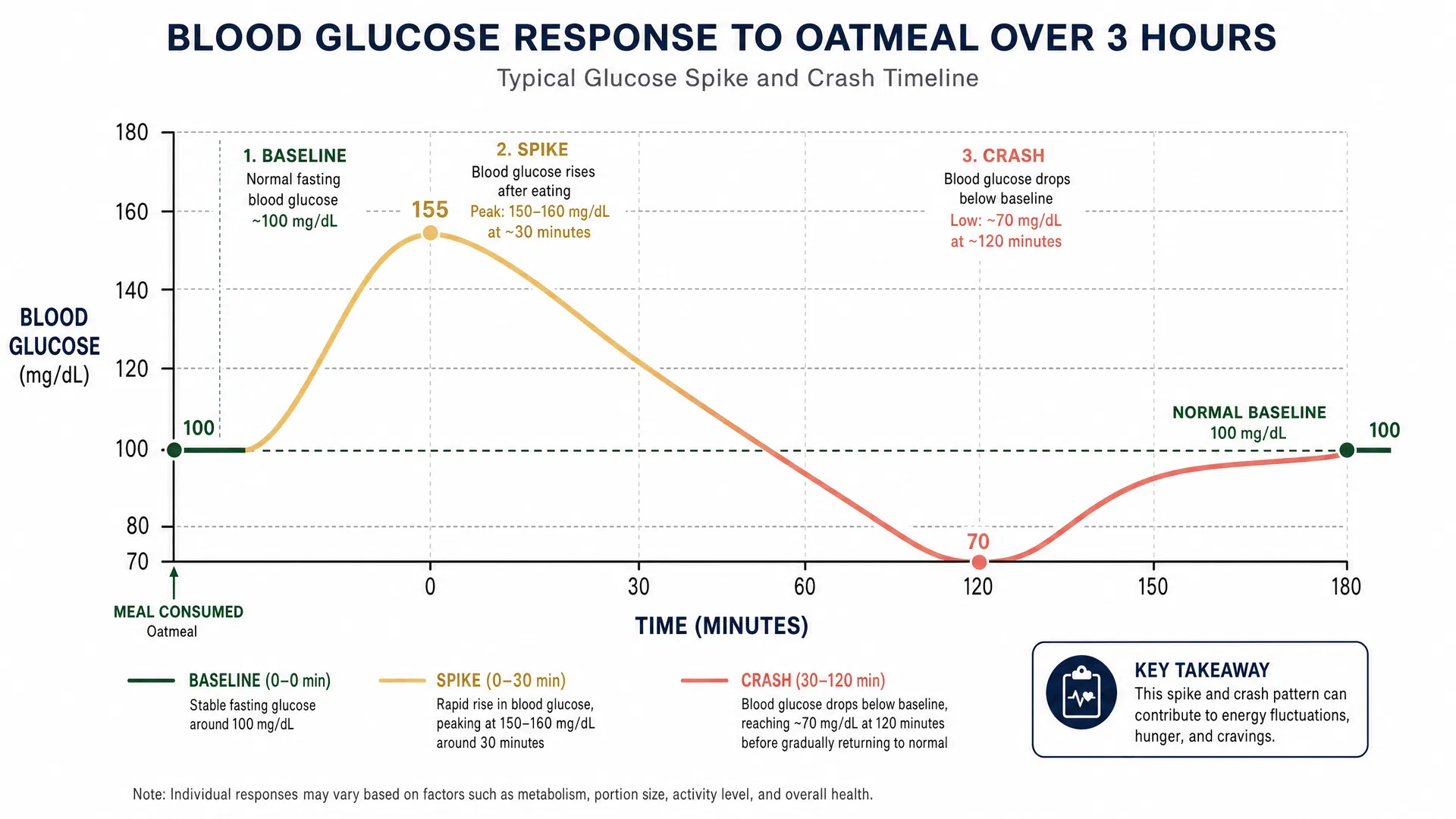
The Glucose Absorption Mechanism
When you consume carbohydrate-containing foods, your digestive system hydrolyzes complex carbohydrates into glucose molecules. This glucose enters the bloodstream, triggering a rapid increase in blood glucose concentration—the postprandial glucose response.
According to research published in the American Journal of Clinical Nutrition (2020), a single serving of oatmeal (approximately 50 grams of carbohydrates) produces a glucose spike of 40-60 mg/dL in healthy, non-diabetic individuals within 15-30 minutes of consumption. This represents a 50-100% increase from baseline glucose levels in many individuals.
Insulin Secretion and Glucose Clearance
In response to elevated blood glucose, the pancreatic beta cells secrete insulin. Insulin binds to insulin receptors on muscle, adipose, and liver cells, facilitating glucose uptake. Under normal circumstances, this system maintains glucose homeostasis within a narrow range (70-100 mg/dL fasting, <140 mg/dL postprandial).
However, the modern diet—characterized by frequent carbohydrate consumption throughout the day—creates a chronic state of postprandial hyperglycemia. A typical "healthy" diet consisting of oatmeal breakfast (80-100g carbs), whole-wheat sandwich lunch (60-80g carbs), and brown rice dinner (50-70g carbs) produces 190-250 grams of daily carbohydrate intake, resulting in 8-12 significant glucose spikes per day.
Part 2: Insulin Resistance and the "Starving Brain" Paradox
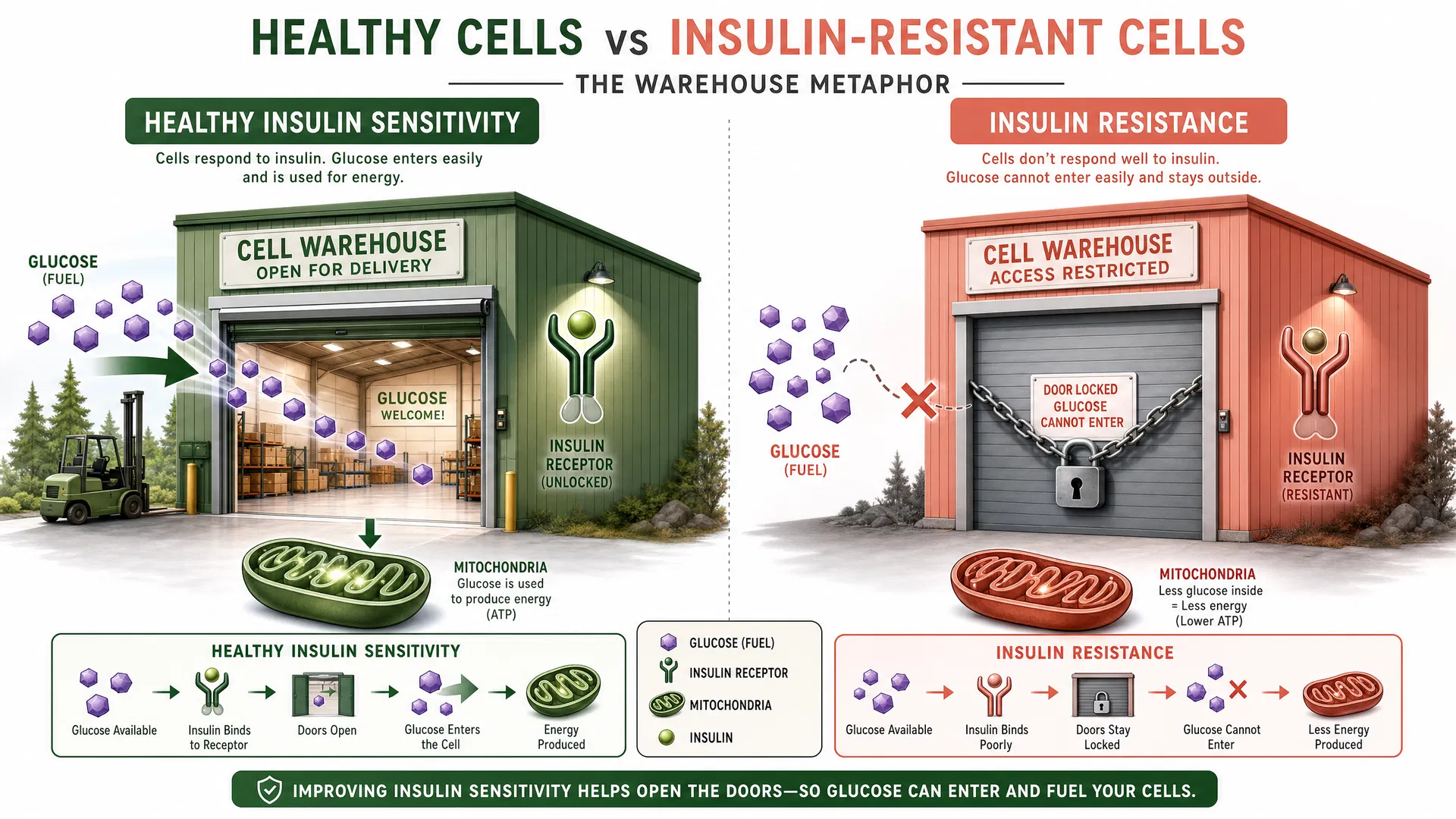
Mechanisms of Insulin Resistance
Chronic postprandial hyperglycemia triggers a compensatory increase in fasting insulin levels. Over time, repeated exposure to elevated insulin concentrations leads to desensitization of insulin signaling pathways—a phenomenon termed insulin resistance.
A landmark study published in JAMA Neurology (2021) demonstrated that insulin resistance impairs glucose uptake in the prefrontal cortex and hippocampus—brain regions critical for executive function, attention, and memory. Notably, this impairment occurs despite normal or elevated blood glucose concentrations.
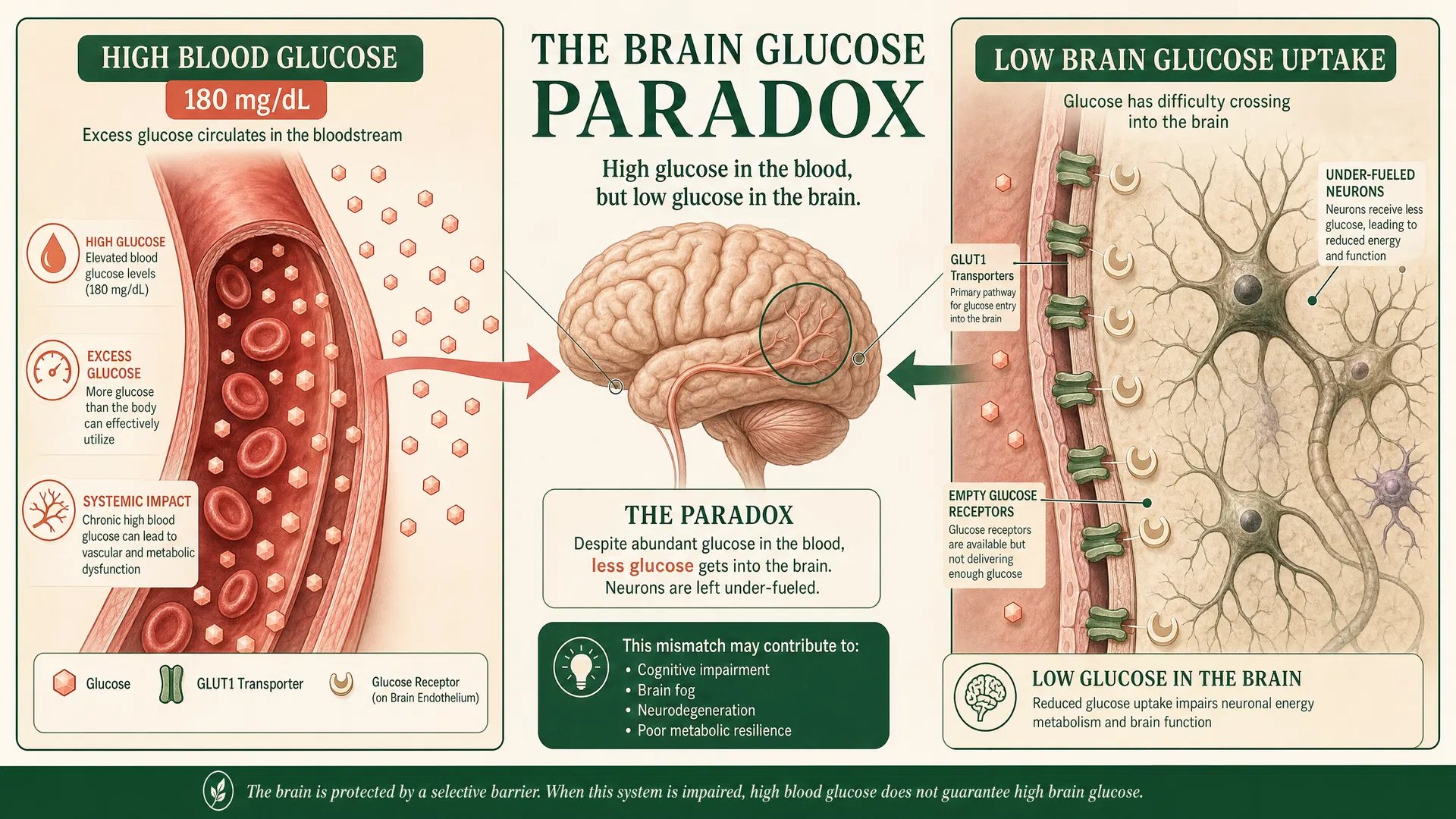
The Neurological Consequences
The brain accounts for approximately 20% of total body energy expenditure and relies almost exclusively on glucose for fuel. Impaired glucose uptake in the context of elevated blood glucose creates a paradoxical state: the brain experiences energy deprivation while the bloodstream is overflowing with glucose.
Research in JAMA Neurology (2022) quantified this effect, demonstrating that individuals with insulin resistance show 30-50% reduction in prefrontal cortex glucose uptake despite normal blood glucose levels, impaired cognitive performance on executive function tasks, and increased subjective fatigue and difficulty sustaining attention.
Part 3: Advanced Glycation End-products (AGEs) and Accelerated Aging
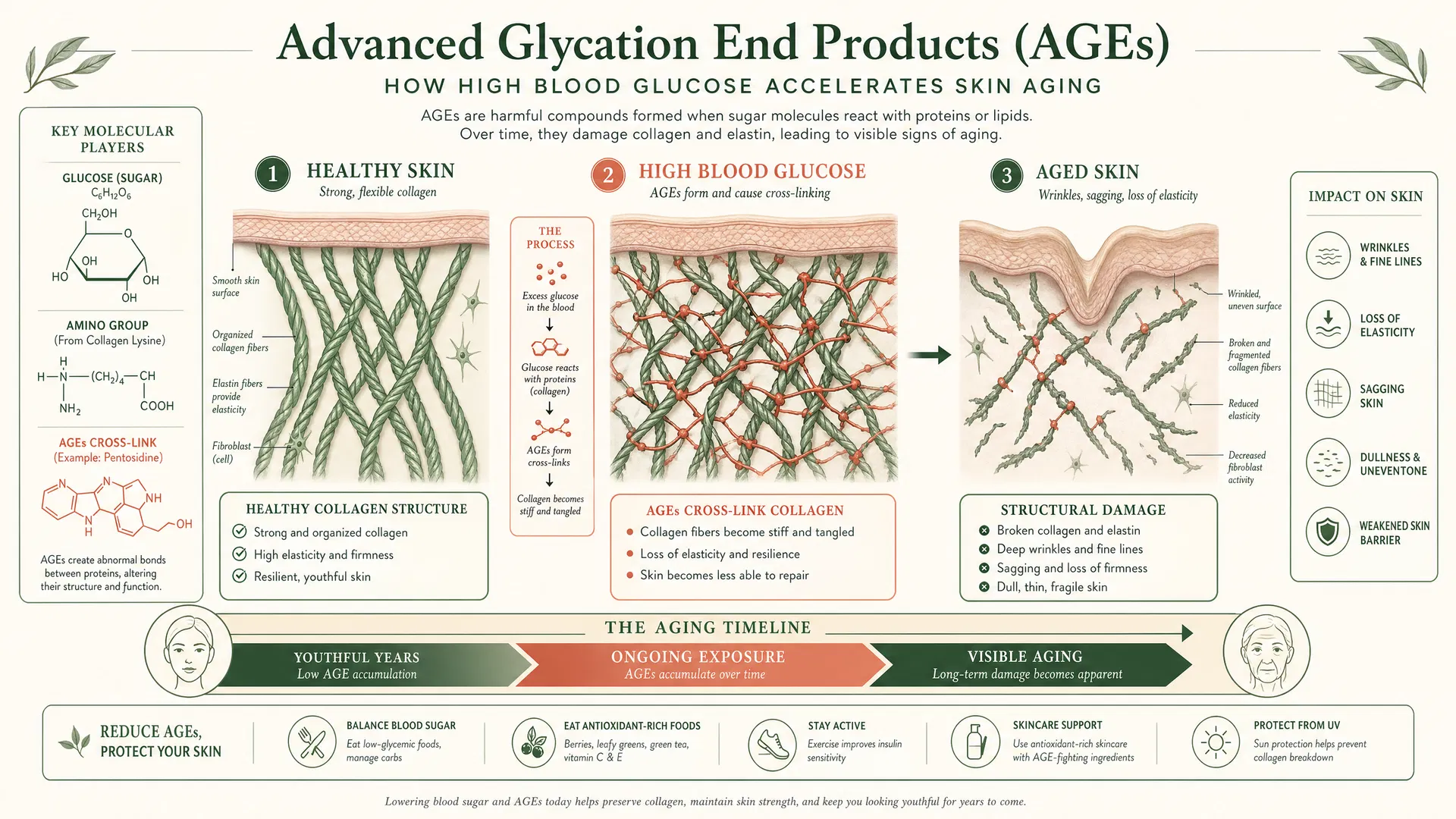
The Glycation Process
When glucose remains in the bloodstream at elevated concentrations for extended periods, it undergoes non-enzymatic glycation—spontaneous binding to amino groups on proteins and lipids. This process generates Advanced Glycation End-products (AGEs), irreversible protein modifications that accumulate with age and chronic hyperglycemia.
Research published in Dermato-Endocrinology (2018) quantified this effect: individuals with chronic postprandial hyperglycemia show 40% more collagen cross-linking compared to those with stable glucose levels, 35% reduction in skin elasticity through AGE-induced elastin cross-linking, and accelerated biological skin aging by 3-5 years.
Systemic AGE Accumulation
AGEs accumulate not only in skin but throughout the body: in joints (accelerating osteoarthritis by 3-5 years), vasculature (increasing arterial stiffness and hypertension), kidneys (contributing to diabetic nephropathy), and eyes (increasing cataract risk).
Part 4: Cognitive Decline and Long-term Neurological Consequences
Longitudinal Evidence
A 10-year prospective study published in the Journal of Clinical Endocrinology & Metabolism (2021) followed 2,000 non-diabetic individuals and found 28% faster cognitive decline in individuals with high glucose variability compared to those with stable glucose levels, increased dementia risk (each 1 mmol/L increase in glucose variability was associated with 1.07× increased risk), and 15-20% greater hippocampal volume loss in high-variability glucose group.
Mechanisms of Cognitive Decline
The mechanisms linking glucose variability to cognitive decline include mitochondrial dysfunction (extreme glucose fluctuations impair mitochondrial function in neurons), neuroinflammation (chronic hyperglycemia activates microglial cells), and amyloid-beta accumulation (impaired glucose metabolism impairs clearance of amyloid-beta).
Part 6: Continuous Glucose Monitoring as a Personalized Biohacking Tool

CGM Technology and Real-time Feedback
Continuous Glucose Monitoring (CGM) systems measure interstitial glucose levels every 5-15 minutes and transmit data to a smartphone application. This real-time feedback enables personalized food response mapping, meal sequencing optimization, exercise timing, and sleep and stress monitoring.
A randomized controlled trial in the American Journal of Clinical Nutrition (2020) examined 100 healthy, non-diabetic individuals using CGM for 4 weeks and found 35-40% decrease in glucose coefficient of variation, 72% reported improved afternoon focus and reduced energy crashes, average 3-5 kg weight loss without calorie restriction, and 68% reported improved sleep quality.
Recommended Resources
Continuous Glucose Monitoring Systems
Educational Resources
Metabolic Support Supplements
Frequently Asked Questions
Do I need to be diabetic to use CGM?
No. CGM is increasingly used by non-diabetic individuals and athletes for performance optimization and longevity. Research demonstrates significant benefits for non-diabetic populations.
How long should I wear a CGM?
Most experts recommend 4-8 weeks to identify patterns and optimize dietary strategies. After initial optimization, periodic use (1-2 weeks per quarter) can monitor ongoing metabolic health.
Can insulin resistance be reversed?
Yes. Studies show that individuals who reduced glucose spikes by 40% over 12 weeks showed significant improvements in insulin sensitivity. Complete reversal typically requires 3-6 months of consistent dietary optimization.
Medical Disclaimer
This article is for educational purposes and does not constitute medical advice. Always consult with a qualified healthcare provider before making significant dietary changes or using medical devices. Individual results vary based on genetics, lifestyle, and underlying health conditions.